Recently, the enterprise of Beidou Xingtong and Xinxingtong recently released a new generation of ultra-small full-system multi-frequency and high-precision RTK positioning module UM960, which provides high-fixed rate, high-precision and reliable GNSS positioning data for intelligent robots, deformation monitoring, unmanned aerial vehicles, handheld GIS and other fields.

The size of the UM960 is 12mm16mm2.6mm, which can bring greater freedom and flexibility to the customer's hardware design. While reducing the difficulty of integration, it can make the terminal product design more integrated and smaller in size, so as to expand to more application scenarios.
Compared with the previous generation of star products, the UM960 cloth plate area has decreased by 84%, and the smaller cloth plate area can also bring cost savings to customers.
The UM960 independently developed by Hexinxingtong supports Beidou, GPS, Galileo, GLONASS and QZSS navigation systems, and the frequency points support BDS B1I/B2I/B3I/B1c/B2a *, GPS L1/L2/L5, Galileo E1/E5b/E5a, GLONASS G1/G2, QZSS L1/L2/L5. The support of multi-frequency points in the whole system enables the UM960 to obtain more available satellites and observations under the same occlusion conditions, improve its data availability under severe occlusion scenarios, improve the fixed rate, and meet the application requirements of various scenarios in different regions.
Thanks to the powerful chip performance of Nabulas IV, the overall performance of UM960 has been significantly improved. The UM960 floating-point computing power has been increased by 5 times, the module computing power has been greatly improved, and the RTK solution time has been greatly shortened; In terms of data output, it supports single point positioning+RTK positioning data 20HZ stable output. In addition, UM960 supports 1408 channels to meet the expansion needs of current and future frequency points.
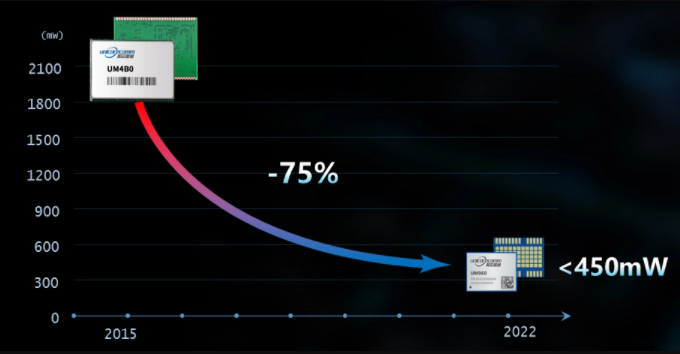
The UM960 ensures that the power consumption is less than 450mW in the case of small size, which is 75% lower than the previous generation of products, and can better meet the industrial application requirements with strict power consumption requirements. The low power consumption feature of UM960 is conducive to solving the problem of working hours of handheld portable devices and other industrial products. In deformation monitoring and other industries, low power consumption can help customers reduce costs.
UM960 supports independent tracking and acquisition of each frequency point. When a certain frequency point is interfered, other frequency points still work normally to ensure that UM960 module can still locate normally in the interference environment.
The UM960 interference detection function can visually display the intensity of interference signals in the current environment. In the performance UAV industry, the environment can be detected before the flight performance to effectively prevent the occurrence of flight accidents; When mapping the location of CORS stations, it is necessary to confirm whether the environment is clean. The UM960 interference detection function can easily detect whether there is interference in the environment, effectively shorten the initial location time of CORS stations, and save the cost of site selection and station construction.
Based on its excellent performance, UM960 can also solve the application pain point problem of low fixed rate and poor accuracy in severe occlusion scenarios, and provide accurate and reliable positioning data for terminal products.

 小任班长的博客
小任班长的博客 






